
And Then There Were Two!
ALPHA particles
It may seem that we are pounding the alpha particle.
Well in a way that is true.
BUT WHY?
WELL,
because there are two different alpha bonds,
 and
and
because the key to understanding the nucleus is understanding
the alpha particle. By the end of this section you will
understand the importance of two types of alpha bonds.
Ever since the first nuclear models were considered, physicists have tried to determine the role of alpha particles IN the nucleus. This is because;
- First, for lite nuclei the binding energy peaks when the number of nucleons in the nucleus is evenly divisible by alpha particles.
- Second, in heavy nuclei the dominant decay mode for nucleons is via the alpha particle rather than by individual protons or neutrons.
- Third, the alpha is extremely stable and a very tightly bonded particle.
AND, Some of the earliest models were based on alpha particles.
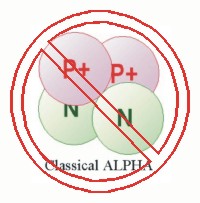
The classical alpha is portrayed in the form of a tetrahedron with the nucleons at the four vertices of that tetrahedron. This configuration makes all the nucleons nearest neighbors. This implies that the alpha particle is held together solely by the strong force because in this configuration the electrical force is pushing the two protons apart.
The tetrahedral form cannot be justified as a correct model when the effects of quarks are considered.
The quark's electrical changes in the protons and neutrons effect the basic configuration of the alpha particle. For brevity the proton neutron quark alpha will be referred to simple as the quark alpha. The image shift of the alpha particle from a tetrahedron to a hexagonal prism is of primary importance for understanding this model and the nucleus. Thus, this fact will be emphasized in several different ways in this paper. It is the hexagonal prism shape that places the quarks, which are confined to the protons and neutrons, in an optimal position that makes the alpha such a stable particle.
This is because in the quark configuration the strong force, color force, the electrical force and the magnetic force are all coupled optimally in the quark alpha particle.
The primary features of the quark alpha are;
- First, each nucleon is made of three quarks, a proton is composed of two up quarks with a +2/3 charge and one down quark with a -1/3 and the neutron that is composed of two downs quarks and one up quark.
- Second, three particles always form a triangle in a single plane, not a sphere.
- Third, each quark in addition to color confinement by the gluon field have electric and magnetic fields that align to provide structure in the nucleus.
Rather than view these nucleons as triangular particles with charged corners, nucleons, protons and neutrons, will be presented in this paper as three quark particles connected by arrows representing nearest neighbor connections and interacting forces. There are the color gluon forces, the electrical forces and magnetic dipole forces.
Thus the figure below is the general form of the ALPHA that will be used throughout this paper.
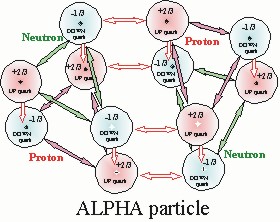
BUT Then There Were Two! WHAT? --- 2 ALPHA particles?
There are two forms of the quark alphas. They are distinguished by their bonding arrangement. These two forms are differ only via their bond arrangment. What has been described to this point is the alpha-1 bond. The alpha-2 bond is created by rotating the nucleons, protons and neutrons, in the alpha by 60 degrees.
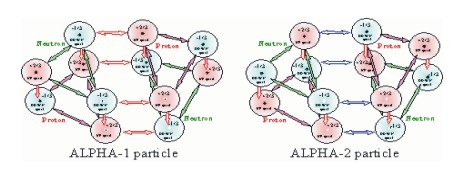
The result of these two bonds are that the nucleus forms a hexagonal lattice structure.
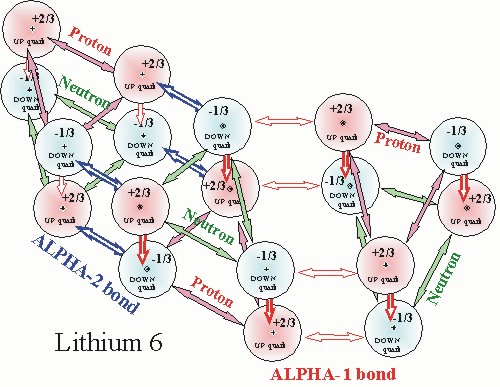
The alpha 1 bond places the two up quarks of the proton adjacent to the two down quarks of the neutron. This is true for both the side by side alignment of nucleons and top to bottom alignment of nucleons in the alpha particle.
The alpha-2 bond places up and down quarks of the proton adjacent to down and up quarks of the neutron. This is again true for both the side by side alignment of nucleons and top to bottom alignment of nucleons in the alpha particle.
In both of these arrangements maximum coupling is achieved for the gluon field, the electric field and the magnetic field. The illustrations show how all these fields work together to make the alpha particle stable and strong. There does not appear to be any method to differentiate between the two types of alphas. However, there should be twice as many alpha 2 bonds as alpha 1 bonds in the nucleus.
Remember both fundamental forces, the strong and EM are manifest in three way in the nucleus. It is the interplay of these three field of those forces that control the structure of the Alpha, which ultimately controls the structure of the nucleus.
The color field or gluons confine the quarks within the nucleons and hold the nucleons within the nucleus.
The electric fields of the quarks within the proton are mirrored by opposite polarity quarks in the neutron. This permits the exact match up of electric charges that are the dominant force controlling the hexagonal lattice nuclear structure.
The magnetic field is the result of the magnetic dipoles of the quarks. Like all magnetic dipoles the dipoles align north to south with each other to form confined magnetic loops within the alpha which results in a zero magnetic dipole for the alpha particle. We know these magnetic alignments in the nucleus are very stable because they are constant and cannot be altered except to precess or align the whole nucleus within a magnetic field. The nuclear magnetic dipole within the nucleus does not change which implies very rigidly positioned quarks!
The model of Lithium 6 below, demonstrates how the two alpha bonds work together to form the interlocking hexagonal lattice structure of the nucleus.
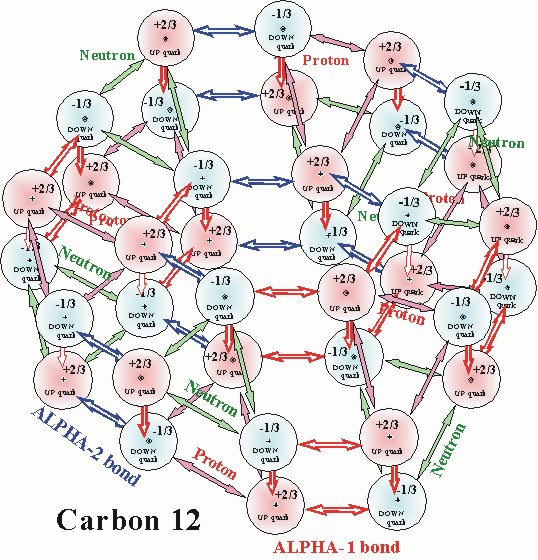
The model of Carbon 12 below further demonstrates how the two alpha
bonds
work together and form the interlocked hexagonal lattice structure
of the nucleus.
The hexagonal carbon ring is another basic structure in the nucleus.
This illustration is difficult for some to visualize so
two different angled photos
of a ZOME Model are shown below this illustration.
|
The model shown here is constructed using nodes and struts obtained from �ZOME�. The nodes in this model represent quarks and the struts represent forces, either color or electromagnetic that hold the three quarks together to form a nucleon. 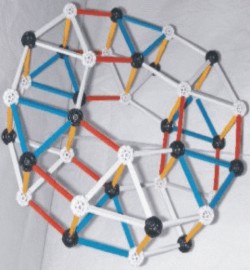
Red struts are alpha-1 bonds white struts in places 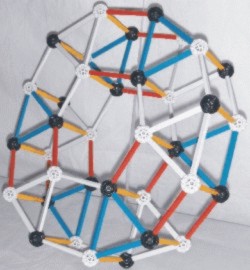
CARBON12 NUCLEUS
LITHIUM6 NUCLEUS 
|
|
|
The foundation particles are quarks represented by �ZOME� nodes. |
|
 |
The black �ZOME� nodes represent �UP� quarks. |
 |
The white �ZOME� nodes represent �DOWN� quarks. |
|
The protons and neutrons are the primary components of the nucleus and both nucleons are composed of three quarks. |
|
|
The first nuclear components, protons and neutrons, are represented in the �ZOME� model by: |
|

|
The proton is shown as a �ZOME� construct using three blue struts (representing the color force) to connect two black nodes (representing the UP quarks) and a white node (representing the DOWN quark). Three blue struts connect black and white nodes to represent
protons. |
 |
The neutron is shown as a �ZOME� construct using three white struts (representing the color force) to connect two white nodes (representing the DOWN quarks) and a black node (representing the UP quark). Three white struts connect white and black nodes to represent
neutrons. |
|
The data and information depicted in and on this web site are covered by © “copyright”. The data and/or information presented in this web site may be used in part or whole if the user gives credit to the authors and references this web site.
|
|